Bomb Radiocarbon Dating of Hawksbill Sea Turtle Carapaces
By: Delaney Reynolds, SRC Intern
After the 1941 attack on Pearl Harbor, Hawai’I, the United States declared martial law. In the decades that followed, the island of Kaho‘olawe, Hawai’I, an island considered sacred to Hawai’ian Natives, was used as a training ground and bombing range for the United States’ Army. Decades of bombing have not only created a threat for human health, but also any wildlife that lived in proximity to the sites. By the official end of the bombing in 2004, the army had detonated over 28,600 from the island and along the shoreline (MacDonald). One marine species that is of particular interest is the critically endangered hawksbill sea turtle (Eretmochelys imbricate).
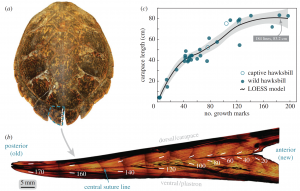
Figure 1: This figure displays a carapace, as well as the correlation between carapace growth and number of growth marks on a scute. Van Houtan, Kyle S, et al. Time in Tortoiseshell: a Bomb Radiocarbon-Validated Chronology in Sea Turtle Scutes. Royal Society Publishing, 27 Nov. 2016.
Sea turtles’ shells are a plate-like structure comprised of “scutes” made of keratin, a component makes up human hair (Van Houtan 2016). Their shells are extremely tough and provide protection from predators. Bomb radiocarbon dating can also be used on shells to estimate growth and maturity and it is data like this that can aid in assessing population status. Hawksbill sea turtles happen to have an extremely reliable chronology when their carapace is carbon dated and can thus tell us their population status before, during, and after the bombing events on Kaho‘olawe to determine how their maturity might have changed or been effected by any radiation.
It has been found that the growth rate of sea turtles varies based on their age. As juveniles, their carapace may grow 4-5 centimeters per year. By age 10, their carapace growth declines to a rate of about 2 centimeters per year. By age 30, the carapace growth then declines to a rate of approximately 1 centimeter per year (Zug).
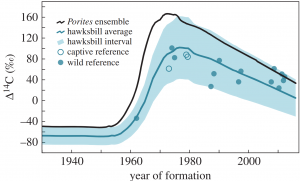
Figure 2: This figure displays the correlation of the percentage of Carbon-14 in the Porites (coral) cores and hawksbills’ shells to the year of core or shell formation. Van Houtan, Kyle S, et al. Time in Tortoiseshell: a Bomb Radiocarbon-Validated Chronology in Sea Turtle Scutes. Royal Society Publishing, 27 Nov. 2016.
Scientists from the national Oceanic and Atmospheric Administration (NOAA) sampled 36 hawksbills of “all life stages, several Pacific populations and spanning eight decades (Van Houtan 2016).” Growth lines were counted and referenced to Carbon-14 values from ascribed coral (Porites) cores. Annually, hawksbills create eight growth lines and it is the distance between each that can be used to estimate age.
In the 1960s, the bombing in Kaho‘olawe began to pick up and this increase can be seen in hawksbill shells’ percentage of Carbon-14 in Figure 2. At the end of the 1970s, the bombing’s demise began as Native Hawai’ian protesters risked their lives to garner the attention of their government. The bombing came to a complete halt in 1993 when Congress voted to end all military use of Kaho‘olawe and transferred the island from martial law back to the state (MacDonald 1972). This decrease in bombing can also be seen in the hawksbill shells’ percentage of Carbon-14 in Figure 2. This tells us that using the bomb radiocarbon dating of hawksbills’ carapaces is an accurate tool for aging. It was determined that wild hawksbills reach maturity between 17 and 22 years of age and captive hawksbills will reach maturity in approximately 12.3 years (Van Houtan 2016).
These bomb radiocarbon dating values also propose a decline in hawksbill trophic status. The same increase and decrease in Figure 2 appear in the coral cores. This may suggest that coral losses may lead to the degradation of sea turtle populations. It has been observed that hawksbill population size is linked to reef structure, but further study is necessary to know for sure.

Figure 3: Hawksbill Sea Turtle “Hawksbill Sea Turtle.” Wikipedia, Wikimedia Foundation, 6 Oct. 2018, en.wikipedia.org/wiki/Hawksbill_sea_turtle.
Hawksbill sea turtles were distinctively oppressed in the historic global tortoiseshell trade because their carapaces contained an abundant amount of structurally aesthetic keratin. Their shell has historically been used to craft jewelry and illegally sold (National Park Service 2017). They were listed as critically endangered by the IUCN in 1996 and placed under the protection of the Federal Endangered Species Act in 1973. In 1991, the State of Florida also passed the Marine Turtle Protection Act prohibiting the “take, possession, disturbance, mutilation, destruction, selling, transference, molestation, and harassment of marine turtles, nests or eggs (Marine Turtle Protection, Florida Fish and Wildlife Conservation Commission).” According to a 2013 report from the National Marine Fisheries Service and U.S. Fish and Wildlife Service, hawksbill populations in the United States seen both declines and recoveries in the last 20 years, but in many cases population trends were unknown.
In Hawai’i, to aid in recovery, the National Park Service has created the Hawai’i Island Hawksbill Turtle Recovery Project to monitor and protect the hawksbills and their nesting habitats found on all Hawai’ian Islands. Acting as predators to mollusks and prey to shark species, sea turtles help to keep the food web in balance and populations in check. As they are a critically endangered species and become increasingly more vulnerable to plastic pollution, consuming plastic grocery bags that resemble jellyfish for example, it is absolutely crucial for us to protect precious sea turtles and their nesting habitats.
Works Cited:
“Hawksbill Sea Turtle.” Wikipedia, Wikimedia Foundation, 6 Oct. 2018, en.wikipedia.org/wiki/Hawksbill_sea_turtle.
“Honu’ea (Hawaiian Haksbill Turtles).” National Park Service, U.S. Department of the Interior, 6 July 2017, www.nps.gov/havo/learn/nature/turtles.htm.
MacDonald, Peter. “Fixed in Time: A Brief History of Kahoolawe.” CORE, 1972.
“Marine Turtle Protection.” Laws/Regulations/Handbook, Florida Fish and Wildlife Conservation Commission, myfwc.com/wildlifehabitats/managed/sea-turtles/protection/.
National Marine Fisheries Service & U.S. Fish and Wildlife Service (2013). 5-YEAR REVIEW Hawksbill Sea Turtle/Eretmochelys imbricate, https://www.fws.gov/northflorida/seaturtles/Docs/2013_hawksbill_sea_turtle_ESA_5-year_Status_Review_FINAL.pdf
Meylan, Anne B, and Marydele Donnelly. Status Justification for Listing the Hawksbill Turtle (Eretmochelys Imbricata) as Criticallyl Endangered on the 1996 IUCN Red List of Threatened Animals. Chelonian Conservation and Biology, 1999.
Van Houtan, Kyle S, et al. Time in Tortoiseshell: a Bomb Radiocarbon-Validated Chronology in Sea Turtle Scutes. Royal Society Publishing, 27 Nov. 2016.
Zug, George R, et al. Age and Growth of Hawaiian Green Seaturtles (Chelonia Mydas): an Analysis Based on Skeletochronology. NOAA, 20 Aug. 2001.